Rev. FCA UNCuyo | 2026 | 58(1) | ISSN 1853-8665
Plant protection
https://doi.org/10.48162/rev.39.209
Mortality of the Toad Grasshopper (Bufonacris claraziana Saussure) Caused by Beauveria bassiana (Balsamo) Vullemin (Hypocreales: Clavicipitaceae) by Two Inoculation Methods under Field-Cage Conditions
Evaluación de la mortalidad causada por Beauveria bassiana (Balsamo) Vullemin (Hypocreales: Clavicipitaceae) contra la tucura sapo comparando dos procedimientos de inoculación en condiciones de jaulas en el campo
Sebastián Alberto Pelizza 2,
Lucas Emanuel Castelli 3,
Franco Armento 4,
1 Universidad Nacional del Centro de la Provincia de Buenos Aires. Comisión de Investigaciones Científicas (UNICEN-CIC). Instituto Multidisciplinario sobre Ecosistemas y desarrollo sustentable. Campus Universitario Tandil. Paraje Arroyo seco S/N.
2 Universidad Nacional de La Plata Facultad de Ciencias Naturales y Museo (FCNyM-UNLP). Instituto de Botánica Carlos Spegazzini. Calle 53 # 477. La Plata (1900). Argentina.
3 Grupo de Entomología Patagónica (GENTPAT), Instituto Patagónico para el Estudio de los Ecosistemas Continentales (IPEEC-CENPAT-CONICET). Bvd. Brown 2915. Puerto Madryn. Chubut. Argentina.
4 Servicio Nacional de Sanidad y Calidad Agroalimentaria (SENASA). Centro Regional Patagonia Sur. Pasaje San Juan 840, El Maitén. Chubut.
5 Universidad Nacional de La Plata (UNLP). Centro de Estudios Parasitológicos y de Vectores (CEPAVE). CCT La Plata CONICET. Boulevard 120 e/60 y 64 S/N. La Plata (1900). Argentina.
6 Comisión de Investigaciones Científicas de la Provincia de Buenos Aires (CICPBA). La Plata. Argentina.
* ymariottini@ecosistemas.exa.unicen.edu.ar
Abstract
Bufonacris claraziana Saussure (Orthoptera: Tristiridae) is a pest grasshopper species in Patagonia, Argentina. Outbreaks of B. claraziana have gained frequency in recent years. This study compared three infection methods (spraying and baits) on B. claraziana mortality caused by Beauveria bassiana under field-cage conditions in the natural habitat. The study was conducted in Cushamen, a typical outbreak area in Chubut province. Three treatments were tested: Treatment I involved spraying with a fungal suspension (1×108 conidia/ml), treatment II used wheat bran baits with the same suspension, and treatment III used wheat bran baits with added canola oil as a phagostimulant. Mortality rates were monitored every 24 hours for 15 days. Total mortality differed among the treatments (F: 17.58; p: 0.0007). After 15 days, mortality was 60%, 70%, and 100% for treatments I, II, and III, respectively. No mortality was observed in treatments I and II until day 5, while treatment III showed 36.7% mortality by day 5 and reached 100% by day 10. Kaplan-Meier survival curves indicated significant differences in survival among treatments, with the lowest survival in treatment III (log-rank test p<0.05). Wheat bran bait with canola oil (treatment III) was the most effective inoculation.
Keywords: field-cage experience, baits and spraying inoculations, Bufonacris claraziana, Patagonia Argentina, entomopathogenic fungus
Resumen
Bufonacris claraziana Saussure (Orthoptera: Tristiridae) es una plaga recurrente en la Patagonia argentina. Este estudio evaluó la mortalidad causada por Beauveria bassiana en B. claraizana mediante dos métodos de infección (aspersión y cebos) bajo condiciones de semicampo en Cushamen, Chubut. Se realizaron tres tratamientos: I) Aspersión con una suspensión fúngica (1×108 conidios/ml), II) Cebos de salvado de trigo con la misma suspensión, y III) Cebos de salvado de trigo más aceite de canola como fagostimulante. La mortalidad se monitoreó cada 24 horas durante 15 días. La mortalidad total fue diferente entre los tratamientos y el control (F: 17,58; p: 0,0007). La mortalidad de los individuos fue del 60%, 70% y 100% para los tratamientos I, II y III, respectivamente, al final del ensayo. En los tratamientos I y II, no se observó mortalidad hasta el día 5, mientras que el III presentó un 36,7% de mortalidad en el día 5, alcanzando el 100% en el día 10. Las curvas de Kaplan-Meier mostraron diferencias significativas en la supervivencia entre los tratamientos; el tratamiento III tuvo la supervivencia más baja (p<0,05). Estos resultados indicarían que los cebos de salvado de trigo con aceite de canola son el método de inoculación más efectivo.
Palabras clave: experiencia en jaulas de campo, inoculación por cebos y por aspersión,
Bufonacris claraziana, Patagonia Argentina, hongos entomopatógenos
Originales: Recepción: 11/09/2024- Aceptación: 17/10/2025
Introduction
Tristirids (Orthoptera: Tristiridae) constitute an endemic family of South American grasshoppers with ten species distributed across the Patagonian steppe, sub-Antarctic forests, and the Puna biogeographic province in Argentina (4, 7). Among these ten species, only Bufonacris claraziana, commonly known as the “Toad grasshopper,” is economically significant in this country and endemic to the Patagonian biogeographic province (2). This species is large (up to 36.5 mm long), robust, and wingless (6); polyphagous and univoltine, with a long obligatory embryonic diapause that extends for more than a single winter (16, 17, 19). Considered a frequent and significant pest (4), outbreaks have become more frequent in recent years, with particularly intense episodes in Chubut, Santa Cruz, and Río Negro provinces during 2016, 2019, and 2022 (18, 19). Both nymphs and adults typically undertake extensive marching migrations through the Patagonian steppe.
In contrast to global trends, the use of chemical insecticides remains the primary option for grasshopper control in our country, resulting in environmental impact and increasing concern about non-target organisms (1, 5, 11). Patagonia, rich in endemic species, has been categorized as vulnerable with maximum conservation priority (8, 23, 24). Consequently, minimized chemical applications have inspired studies on grasshopper pathogens with biocontrol potential, ranging from microsporidia to protists and fungi (26, 28, 30).
Unfortunately, B. claraziana is not susceptible to the microsporidium Paranosema locustae, a naturalized biocontrol agent causing epizootics in other harmful grasshopper species in the country (14). Entomopathogenic fungi (especially Metarhizium anisopliae and Beauveria bassiana) are the main microorganisms used to control different grasshopper species and locust pests (18, 25). To date, eleven fungal formulations are commercially available against grasshoppers and locusts worldwide (10). Recently, Mariottini et al. (2019) used spray inoculation with three strains of B. bassiana, resulting in nearly 100% mortality of adult B. claraziana. Later, these authors tested wheat bran bait with conidia against younger nymphs, obtaining similar mortality rates (Mariottini et al., 2022). To further evaluate the potential of Beauveria bassiana as a biocontrol agent against B. claraziana, we tested two infection procedures under field-cage conditions in natural habitat. We hypothesized that B. bassiana can effectively control Bufonacris claraziana, and under semi-field conditions, efficacy varies with the inoculation method (spray application vs. conidia baits).
Materials and Methods
The experiment took place in a field located in Cushamen County (42°10’23” S, 70°39’39” W), a common outbreak area of B. claraziana in Chubut. The area is characterized by grassy shrub steppe dominated by Poa, Stipa, Festuca, and Chuquiraga avellanedae (3, 24) and “mallines,” low, wet-meadow grasslands with high forage value (21, 22). The site has short, warm summers and cold, snowy winters with average annual temperature of 8°C. Moderate to strong west winds are frequent, and average annual precipitation is 150 mm (20).
Under field-cage conditions, we evaluated nymph mortality caused by B. bassiana using two different infection procedures: spraying and wheat bran baits, as previously described (17, 18, 25, 27). The B. claraziana individuals used in the experiment were captured as young nymphs with entomological nets in early October 2022, at Cushamen. They were placed in wire-screened cages (45 x 40 x 30 cm) with no floor, allowing grasshoppers to interact with the natural soil and vegetation.
The fungal isolate used (LPSc 1227; GenBank accession number: MG012792) was obtained from the Spegazzini Institute culture collection at La Plata National University. This isolate originally came from cadavers of the South American locust Schistocerca cancellata (Serville) (Orthoptera: Acrididae) collected in nature near La Banda (27°44’07” S, 64°14’ 36” W), Santiago del Estero Province, northwestern Argentina (28). Conidia were obtained from cultures grown on potato dextrose agar medium, incubated for 10 days at 25°C in the dark. Conidia were then harvested using disposable cell scrapers (Fisherbrand™) as described by Pelizza et al. (2020).
Fungal suspensions were made with conidia placed in test tubes containing 0.01% (v/v) polyoxyethylene sorbitan monolaurate (Tween 80™; Merck). Suspensions were adjusted to 1×108 conidia/ml using a Neubauer hemocytometer. Viability was assessed after 24 hours as described by Goettel and Inglis (1997). Germination was considered when the germ tube reached half conidium length. Each assessment was performed in triplicate with 300 conidia under each experimental condition. Three treatments were conducted with three replicates of ten individuals each. Treatment I nymphs were sprayed with a 1×108 conidia/ ml aqueous suspension (17). In treatment II, wheat bran (200 g) was sprinkled with a 1×108 conidia/mL suspension. The bait was placed in the cages for the nymphs to consume, as in Mariottini et al. (2022). Treatment III was similar to treatment II, but with the addition of canola oil as a phagostimulant. Controls had 0.01% (v/v) Tween 80®.
Nymph mortality was recorded every 24 hours for 15 days. Dead grasshoppers with no external mycelium were surface-sterilized by sequentially dipping them in 70% ethanol (10-15 seconds), 0.5% sodium hypochlorite solution (1 minute), and sterile distilled water (1 minute, two consecutive baths) (12). They were then placed in a sterile culture chamber, consisting of a Petri dish (60 mm diameter) with a filter-paper disk periodically moistened with sterile distilled water, and incubated at 25°C in the dark. Mycosis was confirmed by microscopic examination of dead grasshoppers. Total mortality for treatments was compared using one-way ANOVA. Mortality rates at 5, 10, and 15 days were compared using the Kruskal-Wallis test and the median survival time (MST). Survival curves were calculated using Kaplan-Meier analysis. Pairwise comparisons between survival curves were performed with the Log-rank test. All analyses were conducted with Infostat (9).
Results
Mortality recorded at the end of the experiment (After 15 days) was 60%, 70%, and 100% for treatments I, II, and III, respectively. Mortality rates differed among treatments (One-Way ANOVA; F: 17.58; df:3, p: 0.0007), with control showing no mortality. Besides, significant differences in mortality percentages were observed at 5, 10, and 15 days (table 1).
Table 1. Kruskal-Wallis test comparing treatment mortality over 5, 10, and 15 days.
Tabla 1. Prueba de Kruskal-Wallis comparando la mortalidad entre los tratamientos a los 5, 10 y 15 días.
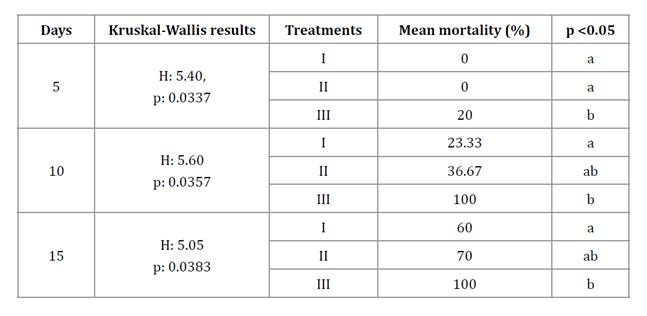
I: Spraying, II: baits of wheat bran plus conidia, III: Baits of wheat bran with conidia plus canola oil. Different letters within each day indicate significant pairwise differences between treatments at p < 0.05.
I: Procedimiento de aspersión, II: Cebos de salvado de trigo con los conidios, III: cebos de salvado de trigo con los conidios más aceite de canola. Letras diferentes dentro de cada día indican diferencias significativas por pares entre tratamientos con p < 0,05.
Up to day 5, treatments I and II evidenced no mortality, whereas treatment III exhibited a significantly higher 36.7 ± 3.3% (p<0.05; table 1). By day 10, treatment III reached 100% mortality, significantly higher than treatment I (p<0.05). Mortality rates were not significantly different between treatment I (36.7%) and II (23.33%).
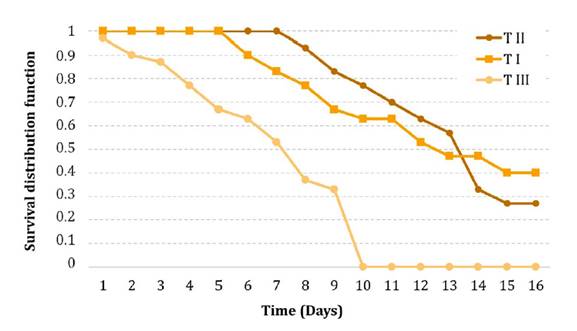
On day 15, mortality in treatment I was similar to that in treatment II (p > 0.05) but differed from treatment III, while treatment II and III were not significantly different from each other (p > 0.05). Kaplan-Meier survival curves (figure 1) further confirmed significant differences among treatments (log-rank test, p < 0.05) (table 2). Treatment III had the lowest survival rate (Log-rank test, p < 0.05).
I: Spraying procedure II: baits of wheat bran plus conidia procedure, III: Baits of wheat bran with conidia plus canola oil.
I: Procedimiento de aspersión, II: Cebos de salvado de trigo con los conidios, III: Cebos de salvado de trigo con los conidios más aceite de canola.
Figure 1. Kaplan-Meier survival curves of Bufonacris claraziana infected with Beauveria bassiana through different inoculation procedures.
Figura 1. Curvas de supervivencia de Kaplan-Meier de Bufonacris claraziana infectada con Beauveria bassiana a través de diferentes procedimientos de inoculación.
Table 2. X2 test for the comparison of the Kaplan-Meier curves of each inoculation procedure.
Tabla 2. Resultados de la prueba log-rank (X²) para la comparación de las curvas de Kaplan-Meier de cada procedimiento de inoculación.
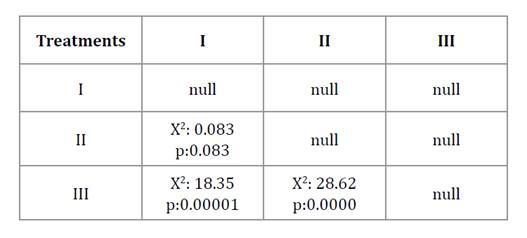
I: Spraying procedure; II: baits of wheat bran plus conidia procedure, III: Baits of wheat bran with conidia plus canola oil.
I: Procedimiento de aspersión, II: Cebos de salvado de trigo con los conidios, III: Cebos de salvado de trigo con los conidios más aceite de canola.
Discussion
According to this study, B. bassiana shows potential for effective biocontrol under field conditions, similar to its performance in controlled laboratory experiments (17, 18). Field-cage effectiveness -where nymphs of B. claraziana were somewhat restricted in their movement and partially protected from environmental factors- suggests that B. bassiana could perform well in actual field scenarios. In real field conditions (i.e., without cages), the use of B. bassiana as wheat bran baits with added canola oil seems promising. Bran baits would offer some protection from UV degradation and other environmental factors, while canola oil would act as a phagostimulant, improving bait acceptance and palatability (15, 27). In general, vegetable oils serve as carriers for control purposes, increasing both attraction to and ingestion of baits, thereby enhancing control efficacy (29, 30). In 2007, Latchininsky et al. (2006), reported that vegetable oils stimulated feeding in Melanoplus sanguinipes, one major grasshopper pest in North America. Ideally, in such a scenario, nymphs to be treated should be the youngest possible, ensuring the highest susceptibility and the shortest time lag until full mortality. Additionally, younger nymphs tend to concentrate, making it easier to control.
In recent years, large areas of Cushamen County have been affected by B. claraziana outbreaks. Most local ranchers are smallholders who rear sheep and goats under subsistence conditions. They manage outbreaks using chemical pesticides like pyrethroids, cypermethrin, organophosphates, dimethoate baits, or mechanical methods like barriers, trenches, and burning of grasshoppers or vegetation (17). These control methods exacerbate species extinction, alter vegetation structure, increase soil erosion, and reduce ecosystem productivity in the Patagonia steppe (21, 24).
Numerous isolates of B. bassiana have been developed worldwide as biocontrol agents for various agriculturally harmful insects (31). While field-cage experiments have their limitations, they provide a valuable step beyond laboratory studies (13). Given the urgent need to revise current control methods for B. claraziana outbreaks, we believe that isolate LPSc 1227 warrants further investigation as a potential management tool for controlling Toad grasshopper populations.
Conclusion
The isolate LPSc 1227 of Beauveria bassiana demonstrated high efficacy against Bufonacris claraziana under field-cage conditions. In our study, wheat-bran baits with canola oil were the most effective inoculation method for early nymphal stages, expanding on previous laboratory results. As discussed, larger-scale open-field trials should continue evaluating the potential of this entomopathogen as a control agent for B. claraziana in Patagonia.
Acknowledgments
We thank Antonio Fermín, production director of the Cushamen Locality.
1. Álvarez, M.; du Mortier, C.; Fernández Cirelli, A. 2013. Behaviour of insecticide chlorpyrifos on soils and sediments with different organic matter content from provincia de Buenos Aires, República Argentina. Water Air Soil Pollution. https://doi.org/10.1007/s11270-013-1453-0.
2. Cabrera, A. L.; Willink, A. 1973. Biogeografía de América Latina. Monogr. Ser. Biol. 13.
3. Cabrera, A. L. 1976. Regiones fitogeográficas argentinas. In: Kugler, W. F. (Ed). Enciclopedia Argentina de Agricultura y Jardinería, II. ACME. 1-85.
4. Carbonell, C. S.; Cigliano, M. M.; Lange, C. E. 2024. Acridomorph (Orthoptera) species of Argentina and Uruguay. http://163.10.203.2/ACRIDOMORPH/
5. Ceccheto, F.; Villalba, A.; Vazquez, N. D.; Ramirez, C. L.; Maggi, M. D.; Miglioranza, K. S. B. 2023. Occurrence of chlorpyrifos and organochlorine pesticides in a native bumblebee (Bombus pauloensis) living under different land uses in the southeastern Pampas, Argentina. Science of the total Environment. https://doi.org/10.1016/j.scitotenv.2023.167117
6. Cigliano, M. M.; Pocco, M. E.; Lange, C. E. 2014. Acridoideos (Orthoptera) de importancia agroeconómica en la República Argentina. In: Roig Juñent, S.; Claps, L.; Morrone, J. J. (Eds). Biodiversidad de Artrópodos Argentinos. Editorial INSUE-UNT. 3: 11-36.
7. Cigliano, M. M.; Lange, C. E. 2019. Toad grasshopper, Bufonacris claraziana (Saussure, 1884) (Orthoptera: Tristiridae). In: Lecoq, M.; Zhang, L. (Eds). Encyclopedia of pest Orthoptera of the World. China Agricultural University Publisher. 235-238.
8. Dinerstein E.; Olson, D. M.; Graham, D. J.; Webster, A. L.; Primm, S. A.; Bookbinder, M. P.; Ledec, G. 1955. A conservation assessment of the terrestrial ecoregions of Latin America and the Caribbean. World Bank.
9. Di Rienzo, J. A.; Casanoves, F.; Ballarin, M. G.; González, L.; Tablada Robledo, C. W. 2011. Grupo InfoStat, FCA, Universidad Nacional de Córdoba. Argentina. http://www.infostat.com.ar
10. Goettel, M. S.; Inglis, G. D. 1997. Hyphomycetes. In: Lacey, L. (Ed). Manual of techniques in Insect Pathology, Academic Press. 213-248.
11. Goldstein, M. I.; Lacher, T. E.; Woodbridge, B.; Bechard, M. J.; Canavelli, S. B.; Zaccagnini, M. E.; Cobb, G. P.; Scollon, E. J.; Tribolet, R.; Hooper, M. J. 1999. Monocrotophos - Induced mass mortality of Swainson’s Hawks in Argentina. 1995-96. Ecotoxicology. (8): 201-214.
12. Inglis, G. D.; Enkerli, J.; Goettel, M. S. 2012. Laboratory techniques used for entomopathogenic fungi: Hypocreales. In: Lacey, L.A. (Ed). Manual of techniques in Invertebrate Pathology. Elsevier. 189-253.
13. Lacey, L. A.; Brooks, W. M. 1997. Initial handling and diagnosis of diseased insects. In: Lacey, L. A. (Ed). Manual of techniques in Insect Pathology. Academic. 1-15.
14. Lange, C. E.; Mariottini, Y.; Plischuk, S.; Cigliano, M. M. 2020. Naturalized, newly associated microsporidium continues causing epizootics and expanding its host range. Protistology. 2020: (14): 32-37. http://dx.doi.org/10.21685/1680-0826-2020-14-1-4
15. Latchininsky, A. V.; VanDyke, K. A. 2006. Grasshopper and locust control with Poisoned baits: a renaissance of the old strategy? Outlooks Pest Management. (17): 105-111.
16. Liebermann, J. 1972. The current state of the locust and grasshopper problem in Argentina. In Proceeding of the international Study Conference on Current and Future Problems of Acridiology. 191-198.
17. Mariottini, Y.; Pelizza, S. A.; Lange C. E. 2019. Susceptibility of Bufonacris claraziana (Orthoptera: Tristiridae) to Beauveria bassiana (Ascomycota: Hypocreales) under controlled conditions. Biocontrol Science and Technology. (29): 817-821. http://dx.doi.org/10.1080/09583157.2019.1597335
18. Mariottini, Y.; Lange, C. E.; Pelizza, S. A. 2022. Laboratory test of Beauveria bassiana (Balsamo-Crivelli) Vuillemin s.l. (Hypocreales: Clavicipitaceae) baits for the biocontrol of the Toad grasshopper pest, Bufonacris claraziana (Saussure) (Orthoptera: Tristiridae). Egyptian Journal of Biological Pest Control. 32:110. https://doi.org/10.1186/s41938-022- 00609-4
19. Medina, H.; Massola, M. P.; Zembo, E.; Verdejo, A.; Shicappacassi, M.; Azzaro, F.; Cienfuegos, M. S.; Sánchez, M.; Armento, F.; Mariottini, Y.; Fernández Arhex, V., Bobadilla, S.; Bobadilla, W.; Binda, S.; Napoli, S.; Camiña, D. 2020. Guía de recomendaciones para el manejo y control de la tucura sapo. SENASA. https://www.argentina.gob.ar/sites/default/files/guiade_recomendaciones_para_el_monitoreo_y_control_de_tucura_sapo.pdf
20.Mendez Casariego, H.; Bran, D. E.; Peralta, C. R.; Madariaga, M. C.; Huerta, G. J.; Villarreal, P.; Oliva, G. E.; López Raggi, F.; Santagni, A.; Easdale, M. H.; Sarmiento, A. C.; Giraudo, C. G.; Rial, P. E.; Escobar, J. M.; Marciani, S. N.; Martinez, R. S.; Elissalde, N. O. 2005. Programa Nacional de Ecorregiones. La región Patagonia: Centros Regionales Patagonia Norte y Patagonia Sur; INTA Centro Regional Patagonia Norte. EEA Bariloche. Comunicaciones Técnicas.
21. Olson, D.; Dinerstein, E. 2002. The Global 200: Priority Ecoregions for Global Conservation. Annals of Missouri Botanical Garden. 89(2): 199-224.
22. Oyarzabal, M.; Clavijo, J. R.; Oakley, L. J.; Biganzoli, F.; Tognetti, P. M.; Barberis, I.; Maturo, H. M.; Aragón, R.; Campanello, P. I.; Prado, D.; Oesterheld, M.; León, R. J. 2018. Unidades de vegetación de la Argentina; Asociación Argentina de Ecología. Ecología Austral. 28: (1-4): 40-63. https://doi.org/10.25260/EA.18.28.1.0.399
23. Paruelo, J.; Aguiar, M. 2003. Impacto humano sobre los ecosistemas. El caso de la desertificación. Ciencia Hoy. 13.
24. Paruelo, J.; Golluscio, R.; Jobbágy, E.; Canevari, M.; Aguiar, M. 2005. Situación ambiental en la estepa patagónica. In: Brown, A.; Martinez Ortiz, U.; Acerbi, M.; Corcuera, J. (Eds). La situación ambiental argentina. Buenos Aires: Fundación Vida Silvestre Argentina. 303-313.
25. Pelizza, S. A.; Mariottini, Y.; Russo, M. L.; Cabello, M. N., Lange, C. E. 2013. Survival and fecundity of Dichroplus maculipennis and Ronderosia bergi (Orthoptera: Acrididae: Melanoplinae) following infection by Beauveria bassiana (Ascomycota: Hypocreales) under laboratory conditions. Biocontrol Science and Technology. (23): 701-710. https://doi.org/10.1080/09583157.2013.791668
26. Pelizza, S. A.; Mariottini, Y.; Russo, L. M.; Vianna, M. F.; Scorsetti, A. C.; Lange, C. E. 2017. Beauveria bassiana (Ascomycota: Hypocreales) introduced as an endophyte in corn plants and its effects on consumption, reproductive capacity, and food preference of Dichroplus maculipennis (Orthoptera: Acrididae: Melanoplinae). Journal of Insect Science. 17(2): 53. https://doi.org/10.1093/jisesa/iex024
27. Pelizza, S. A.; Mariottini, Y.; Russo, L. M.; Vianna, F. M.; Scorsetti, A. C.; Lange, C. E. 2019. Application of Beauveria bassiana using diferent baits for the control of grasshopper pest Dichroplus maculipennis under field cage conditions. Journal of King Saudi University Science. 31(4): 1111-1115. https://doi.org/10.1016/j.jksus.2019.02.014
28. Pelizza, S. A.; Medina, H.; Ferreri, N. A.; Elíades, L. A.; Pocco, M. E.; Stenglein, S. A.; Lange, C. E.; 2020. Virulence and enzymatic activity of three new isolates of Beauveria bassiana (Ascomycota: Hypocreales) from the South American locust Schistocerca cancellata (Orthoptera: Acrididae). Journal of King Saudi University Science. (32): 44-47. https://doi.org/10.1016/j.jksus.2017.11.006
29. Pérez Panti, O.; García de la Cruz, R.; González Hernández, H.; Sánchez Soto, S.; Moscoso Ramírez, P. A.; Izquierdo Reyes, F. 2024. Bio-efficacy of entomopathogenic fungi and vegetable oils against the pink pineapple mealybug: Dysmicoccus brevipes (Cockerell). Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 56(1): 83-93. DOI: https://doi.org/10.48162/rev.39.125
30. Sepúlveda, L.; Pietrantuono, A. L.; Buteler, M.; Fernández-Arhex, V. 2019. Effect of Vegetable Oils as Phagostimulants in Adults of Dichroplus vittigerum (Orthoptera: Acrididae). Journal of Economic Entomology. 112(6): 2649-2654. https://doi.org/10.1093/jee/toz190
31. Sokolova, Y. Y.; Lange, C. E.; Fuxa, J. R. 2006. Development, ultrastructure, natural occurrence, and molecular characterization of Liebermannia patagonica n. gen., n. sp., a microsporidium of the grasshopper Tristira magellanica. Journal of Invertebrate Pathology. (91): 168-182. https://doi.org/10.1016/j.jip.2006.01.003
32. Vega, F. E.; Meyling, N. V; Luangsaard, J. J.; Blackwell, M. 2012. Fungal entomopathogenes. In: Vega, F. E.; Kaya, H. K. (Eds). Insect Pathology. Elsevier. 171–220. DOI: 10.1016/ B978-0-12-384984-7.00006-3